- B.Sc. with Year in Industry, Biochemistry, University of Leicester (UK), 2012
- Ph.D., Cell, Molecular, Developmental Biology, and Biophysics Program, Johns Hopkins University, 2018
- Post-Doc, Montana State University, 2023
MISSION
Determine how: i) bacterial and archaeal CRISPR adaptive immune systems make DNA-based memories of past phage infections, and ii) how CRISPR-generated nucleotide messengers signal an immune response. We’ll repurpose these mechanistic insights to develop medical and biotech applications.
Mechanisms and applications of diverse CRISPR integration complexes
Vertebrates, bacteria, and archaea have domesticated transposases (e.g., RAG1 and Cas1) for adaptive immunity. Transposases, integrases and recombinases often co-opt additional DNA-bending proteins (e.g., IHF, HU, H-NS, or HMGB1) that facilitate DNA integration and excision. However, the structural role of DNA folding during this mobilization of DNA remains largely enigmatic.
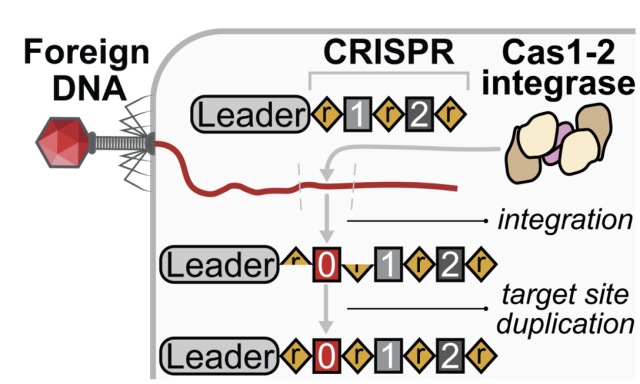
Bacteria and archaea acquire resistance to viruses and plasmids by integrating fragments of foreign DNA into the first repeat of a CRISPR array. Preferential integration at the first CRISPR repeat ensures efficient transcription and processing of CRISPR RNAs that target the most recently encountered genetic parasites.
We recently determined a 560 kDa integration complex structure that explains how Pseudomonas aeruginosa Cas (Cas1-2/3) and non-Cas proteins (IHF) bind conserved DNA sequence motifs upstream of the CRISPR to fold 150 base-pairs of the genome for site-specific integration of foreign DNA.
Click here to watch a video about key findings.
Mechanisms and applications of CRISPR-associated immune signaling pathways
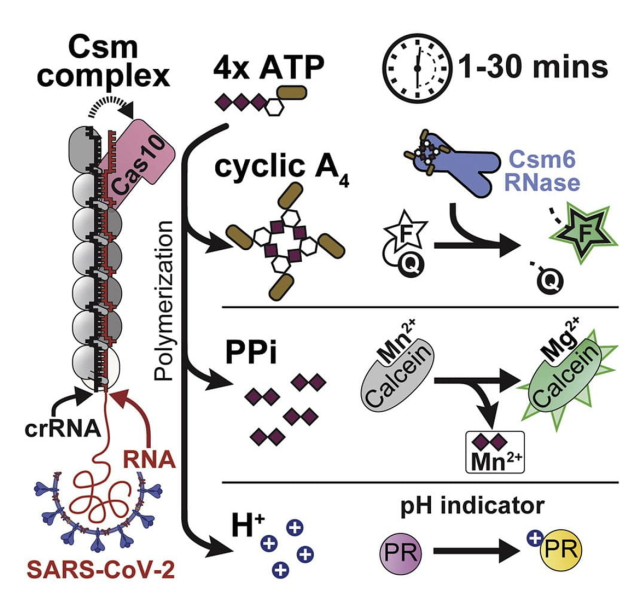
Nucleotides are the most primitive cellular messengers that regulate critical processes across the tree of life, including anti-viral immune responses, cell morphology, and motility. Recent studies show eukaryotic innate immune systems such as cGAS-STING share ancestry with bacterial immune systems. For example, viral RNA-bound Csm and Cmr complexes generate nucleotide messengers (e.g., cA3) that abort infection (e.g., RNA or DNA degradation) by activating immune effector proteins. To address a growing need for rapid and sensitive diagnostics, I co-invented an innovative RNA-guided Csm system for sensitive and sequence-specific detection of SARS-CoV-2 RNA, that repurposes a nuclease immune effector.
Computational “guilt-by-association” analyses suggest that CRISPR-generated immune signaling molecules activate a broad range of effectors, and kick-off diverse biochemical cascades – Some of these immune cascades bear a striking similarity to eukaryotic immune pathways.
See website and reach out to me to discuss current research questions!
- Santiago-Frangos A, Henriques WS, Wiegand T, Gauvin CC, Buyukyoruk M, Graham AB, Wilkinson RA, Triem L, Neselu K, Eng E, Lander GC, Wiedenheft B*.: Structure reveals why genome folding is necessary for site-specific integration of foreign DNA into CRISPR arrays. Nature Structural & Molecular Biology.
- Nemudraia A, Nemudryi A, Buyukyoruk M, Scherffius AM, Zahl TR, Wiegand T, Pandey S, Nichols JE, Hall L, McVey A, Lee HH, Wilkinson RA, Snyder LR, Jones JD, Koutmou KS, Santiago‐Frangos A*, Wiedenheft B*.: Sequence-specific capture and concentration of viral RNA by type III CRISPR system enhances diagnostic. Nature Communications
- Patent on RNA purification and detection using a type III CRISPR complex.
See a more complete summary on my site here.

